Key Takeaway:
- RNA Damage Sunburn research found sunburn inflammation is triggered mainly by RNA damage, not DNA damage, as long believed.
- A protein called ZAK-alpha activates the skin’s rapid stress response after UV exposure.
- Findings could reshape dermatology research and lead to new treatments for sunburn and inflammatory skin diseases.
Scientists from the University of Copenhagen and Nanyang Technological University, Singapore, report that RNA damage, not DNA damage, triggers sunburn inflammation, a discovery published Monday that could reshape skin biology research and future treatments.
Researchers Identify RNA As Primary Sunburn Trigger
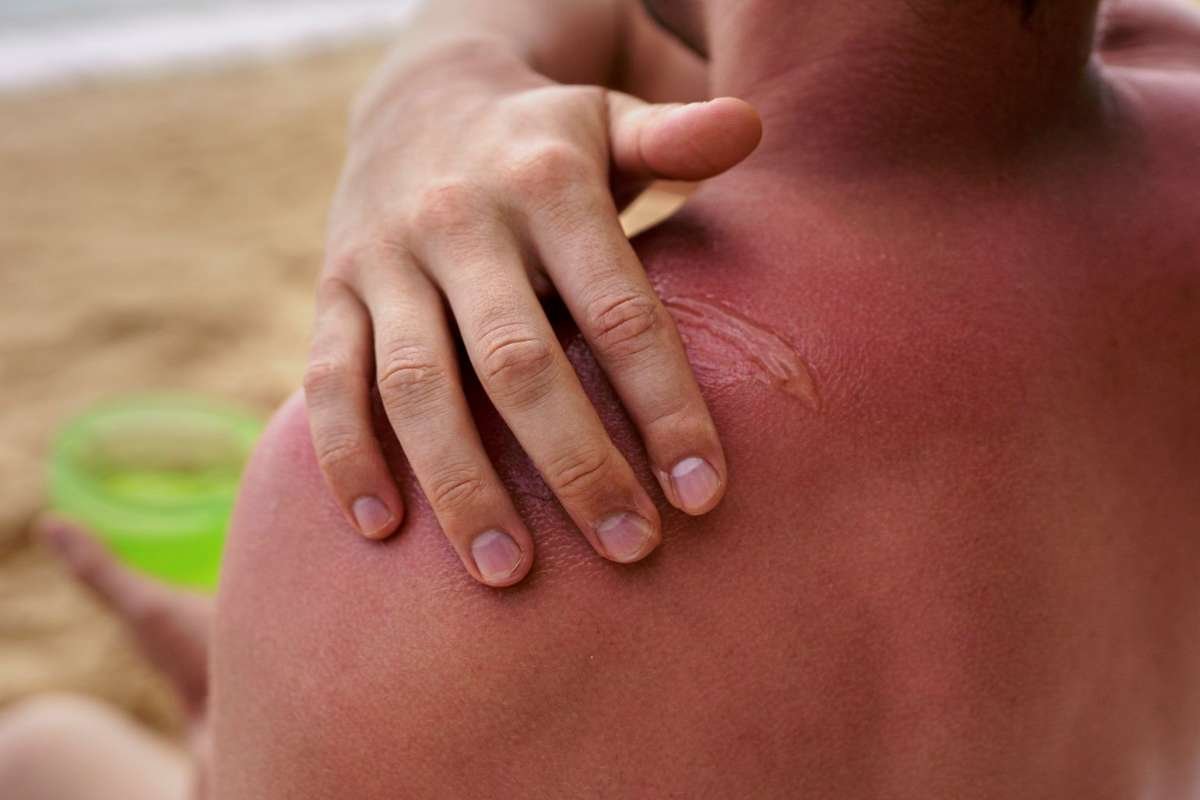
For decades, scientists believed sunburn resulted mainly from RNA damage caused by ultraviolet radiation. New research now suggests the skin’s immediate response to sun exposure begins with injury to RNA molecules instead.
The international study, published in Molecular Cell, shows that ultraviolet radiation first damages messenger RNA, or mRNA, prompting inflammation and cell death associated with sunburn.
RNA Damage Sunburn the DNA, leading to cell death and inflammation. So the textbooks say,” said assistant professor Anna Constance Vind of the University of Copenhagen’s Department of Cellular and Molecular Medicine. “But we were surprised to learn that damage to RNA, not DNA, causes the acute effects of sunburn.”
DNA stores long-term genetic information, while RNA acts as a temporary messenger carrying instructions needed to produce proteins essential for cell function.
Researchers say RNA’s dynamic nature makes it more vulnerable to ultraviolet radiation, triggering faster cellular reactions.
Study Reveals Key Role Of ZAK-Alpha Protein
Scientists conducted experiments using both mice and human skin cells to analyze how skin responds after ultraviolet exposure. They observed similar inflammatory reactions across both models.
The team identified a protein known as ZAK-alpha as central to the process. When RNA damage sunburn occurs, ZAK-alpha activates a “ribotoxic stress response,” signaling cells to halt protein production and initiate inflammation.
“We found that the first thing the cells respond to after exposure to UV radiation is damage to the RNA,” said Professor Simon Bekker-Jensen, a study co-author. “This is what triggers cell death and inflammation of the skin.”
In laboratory mice lacking the ZAK gene, typical sunburn symptoms such as inflammation and cell death did not occur after ultraviolet exposure, confirming the protein’s role.
Bekker-Jensen said the discovery shows skin cells rely on a rapid cytoplasmic stress signal rather than slower DNA-based responses.
Findings Could Transform Skin Disease Research And Treatment
The discovery represents what researchers describe as a major shift in understanding how human skin protects itself from environmental damage.
“The fact that DNA does not control the skin’s initial response to UV radiation is quite the paradigm shift,” Vind said.
While DNA mutations remain linked to long-term risks such as skin cancer, scientists say RNA Damage Sunburn explains the immediate redness, swelling, and pain of sunburn.
Dr. Franklin Zhong, assistant professor at Nanyang Technological University’s Lee Kong Chian School of Medicine and a co-author, said the findings may influence treatment approaches for inflammatory skin conditions worsened by sunlight.
“Understanding how our skin responds at the cellular level to UV damage opens the door to innovative treatments for certain chronic skin diseases,” Zhong said.
Researchers emphasized that traditional sun protection measures, including sunscreen use, protective clothing, and limiting exposure during peak sunlight hours, remain essential.
However, the new evidence suggests future therapies may target RNA damage pathways rather than focusing solely on DNA protection.
“This new knowledge turns things upside down,” Bekker-Jensen said. “Now we need to rewrite the textbooks, and it will affect future research on the effects of UV radiation on the skin.”
Scientists say further studies are needed before clinical treatments emerge, but the findings already challenge one of dermatology’s longest-standing assumptions.