Key Takeaway:
- mRNA cancer vaccines activate multiple immune pathways, not just previously assumed dendritic cells.
- Newly identified cDC2 cells can successfully trigger tumor-fighting CD8+ T cell responses.
- Discovery may improve personalized cancer vaccines and strengthen future immunotherapy treatments.
Researchers at Washington University School of Medicine report that mRNA cancer vaccines activate previously unknown immune pathways involving multiple dendritic cell types, strengthening antitumor responses and opening new possibilities for personalized cancer immunotherapy.
Researchers Discover Alternative Immune Activation Mechanism
Scientists studying next-generation cancer treatments say mRNA vaccines can stimulate powerful antitumor immunity even without immune cells once thought essential, challenging long-standing assumptions in immunology.
The findings, published April 15 in Nature, show that messenger RNA vaccines activate CD8+ T cells. Key cancer-fighting immune cells, through more than one dendritic cell pathway. The discovery could improve therapeutic vaccines targeting tumors such as melanoma, bladder cancer, and small-cell lung cancer.
“For years, we believed one specific dendritic cell population was required to initiate these responses,” said Dr. Kenneth M. Murphy, an immunologist at Washington University School of Medicine and senior author of the study. “Our results show the immune system has built-in redundancy that allows strong protection even when that pathway is absent.”
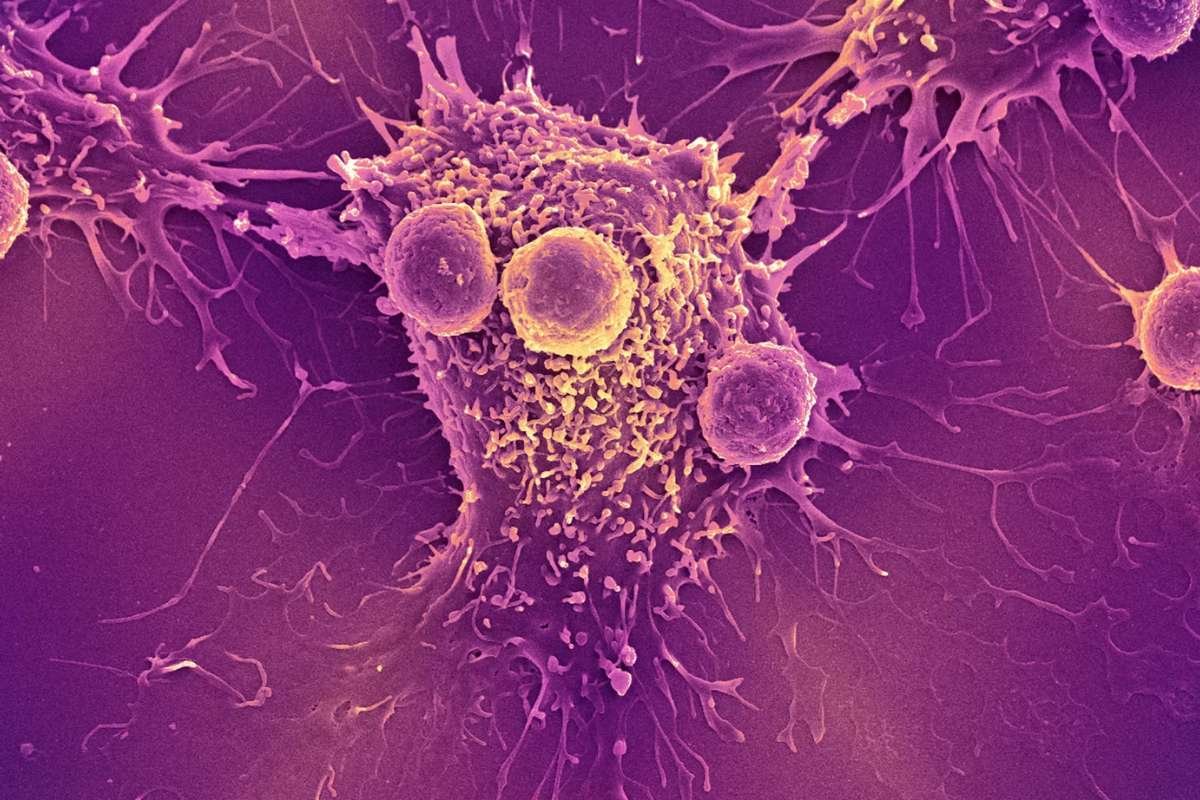
Researchers conducted experiments using genetically engineered mouse models lacking classical type 1 dendritic cells, or cDC1, which traditionally are considered essential for activating CD8+ T cells.
Despite their absence, vaccinated mice mounted strong immune responses capable of eliminating sarcoma tumors, surprising scientists and prompting deeper investigation.
Secondary Dendritic Cells Show Critical Cancer-Fighting Role
The study identified classical type 2 dendritic cells, known as cDC2, as unexpected contributors to tumor immunity. Previously viewed as less important in antiviral or vaccine-driven responses, these cells demonstrated the ability to prime cancer-targeting T cells.
Instead of directly producing tumor antigens from vaccine instructions, cDC2 cells relied on a process researchers describe as “cross-dressing.” Other immune cells first generated tumor proteins and displayed them on their surfaces before transferring antigen complexes to cDC2 cells.
“This mechanism expands how antigen presentation can occur,” said Dr. William E. Gillanders, a surgical oncologist at the Siteman Cancer Center and co-author of the study. “It helps explain why mRNA Cancer Vaccines can remain effective across different biological conditions and patients.”
Scientists say the dual involvement of cDC1 and cDC2 cells may clarify why some cancer patients respond strongly to immunotherapy while others show limited benefit.
Distinct molecular signatures observed in T cells activated by each dendritic subtype also suggest that immune responses could be customized through vaccine design.
Findings May Accelerate Personalized Cancer Vaccines
mRNA Cancer Vaccines work by delivering genetic instructions that prompt immune cells to produce tumor-associated proteins. These proteins are displayed on cell surfaces, allowing CD8+ T cells to recognize and destroy cancer cells.
The new research indicates that engaging multiple immune pathways may strengthen treatment reliability and improve outcomes for cancers historically resistant to immunotherapy.
“This gives vaccine developers new targets for optimization,” Murphy said. “By understanding which dendritic cells are involved, we can design vaccines that generate stronger and more consistent immune responses.”
Experts say the findings arrive as clinical trials expand worldwide testing mRNA-based cancer vaccines as early treatment options and personalized therapies tailored to individual tumors.
Although the experiments were conducted in animal models, researchers believe the mechanisms likely apply to human immune systems and warrant further clinical investigation.
Beyond oncology, the discovery could influence vaccine development for infectious diseases and autoimmune conditions by reshaping how scientists think about immune activation.
The study highlights growing momentum behind mRNA technology following its success during the COVID-19 pandemic, signaling a broader shift toward programmable immunotherapies.
Researchers say future studies will focus on translating the findings into clinical applications and identifying biomarkers that predict patient responses.