A multi-institutional study analyzing nearly 356,000 human hippocampal cells reports ongoing adult neurogenesis and identifies distinct molecular signatures linked to aging, Alzheimer’s disease, and exceptional memory in adults over 80.
Scientists Map Adult Neurogenesis In Human Hippocampus
Researchers report evidence that new neurons continue to form in the adult human hippocampus and that this process changes with aging and cognitive decline.
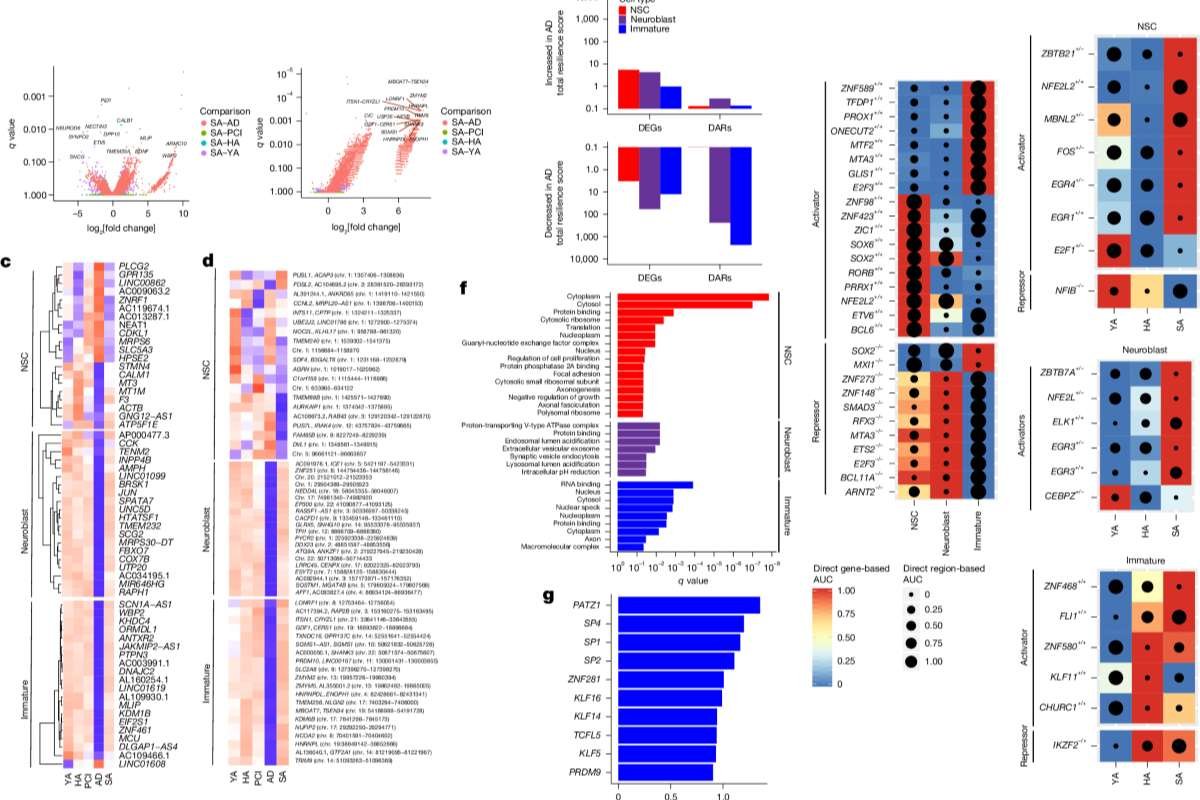
The study, published in the journal Nature, analyzed 355,997 nuclei from post-mortem hippocampal tissue across five groups: young adults, healthy older adults, people with preclinical Alzheimer’s pathology, patients with Alzheimer’s disease, and so-called SuperAgers over age 80 with exceptional memory.
Using single-nucleus RNA sequencing and chromatin accessibility profiling, the team identified neural stem cells, neuroblasts, and immature neurons in the dentate gyrus, a brain region critical for memory.
“We observed a clear developmental trajectory from neural stem cells to mature neurons in the adult human hippocampus,” the authors wrote, adding that gene regulatory networks and chromatin changes distinguish each stage.
The findings address a long-standing debate over whether adult hippocampal neurogenesis occurs in humans. Prior studies produced conflicting results due to differences in tissue processing and analysis methods.
“This work provides strong molecular evidence that neurogenesis persists in adulthood,” said a neuroscientist involved in the research, who was not authorized to discuss unpublished details beyond the paper.
Alzheimer’s Linked To Epigenetic Disruption Of New Neurons
The study found that while neural stem cells were present across all groups, individuals with Alzheimer’s disease showed significantly fewer neuroblasts and immature neurons compared with healthy adults.
Notably, many of the changes were not limited to gene expression. Instead, the most pronounced differences appeared in chromatin accessibility — chemical modifications that influence whether genes can be turned on or off.
“The majority of diagnosis-driven alterations were observed in differentially accessible chromatin regions rather than in gene expression alone,” the researchers reported.
In people with preclinical pathology, individuals who had brain changes associated with Alzheimer’s but no diagnosed dementia, the team detected early epigenetic alterations in neurogenic cells. These changes became more pronounced in patients with confirmed Alzheimer’s disease.
Pathways linked to synaptic plasticity and neurotransmission were among the most disrupted, suggesting that impaired formation and integration of new neurons may contribute to memory decline.
The authors cautioned that the study relies on post-mortem samples and includes a relatively small number of donors, which may limit statistical power. Still, they said the consistency of chromatin-based signatures across groups strengthens the findings.
SuperAgers Show Distinct ‘Resilience’ Signature
Adults classified as SuperAgers — individuals 80 or older whose episodic memory performs at levels comparable to people decades younger — displayed a different molecular profile.
Compared with both healthy older adults and Alzheimer’s patients, SuperAgers showed higher numbers of immature neurons and distinct patterns of chromatin accessibility in neuroblasts and immature neurons. Findings linked to preserved adult neurogenesis.
The researchers described this as a “resilience signature,” marked by preserved or enhanced regulatory networks associated with neuronal differentiation and synaptic function.
In addition, analyses of CA1 neurons and astrocytes — key hippocampal cell types — revealed gene and chromatin patterns linked to preserved synaptic integrity in successful aging.
“Preservation of excitatory synapse integrity appears to be a hallmark of healthy cognitive aging,” the study concluded.
The researchers said identifying these molecular networks could help guide future therapeutic strategies aimed at maintaining cognitive function during aging or slowing the progression of Alzheimer’s disease.
They emphasized that larger studies will be needed to confirm the resilience signatures and determine whether targeting specific gene regulatory networks can alter disease trajectories.