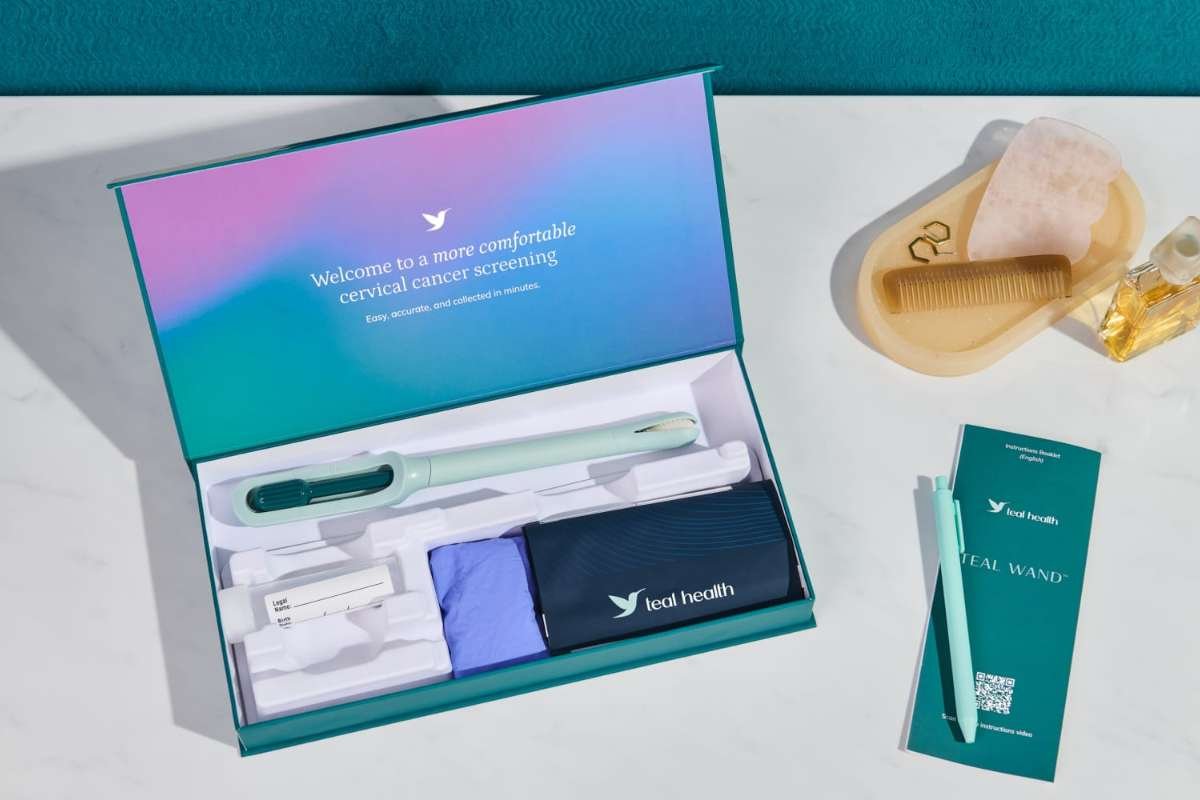
The Food and Drug Administration has cleared an at-home HPV self-collection kit, enabling patients to screen for cervical cancer from home with an at-home HPV test, aiming to improve early detection and access for underserved populations.
FDA Decision Expands Screening Access
U.S. regulators on Tuesday cleared the Onclarity HPV Self-Collection Kit for at-home use and approved the BD Onclarity HPV Assay with extended genotyping. The decision is intended to remove barriers for people who do not undergo routine cervical cancer screening.
Cervical cancer is largely preventable through early detection of human papillomavirus, or HPV, which causes nearly all cases. Health officials estimate that about 60% of cases occur in individuals who are unscreened or under-screened.
“Expanding access to screening is one of the most important steps we can take to prevent cervical cancer,” said Jeff Andrews, vice president of medical affairs at Waters Corporation. “At-home HPV self-collection is a game changer for making screening easier to complete.”
Public health experts say self-collection tools can help reach patients who face barriers such as limited clinic access, stigma, or time constraints. The FDA clearance reflects growing efforts to improve screening participation and reduce cancer deaths.
Advanced Testing Improves Early Detection
The At-home HPV test works with the BD Onclarity HPV Assay, which detects high-risk HPV types linked to cervical cancer. The test identifies six individual genotypes and three grouped categories, providing detailed risk information for clinicians.
Samples collected at home are processed using the BD COR System, an automated platform that prepares, analyzes, and reports results while maintaining sample integrity. The system uses robotics to streamline laboratory workflows and reduce errors.
The product’s performance was evaluated in collaboration with the National Cancer Institute through its cervical cancer “Last Mile” initiative, SHIP Trial. Researchers assessed the accuracy of self-collected samples compared with traditional clinical methods.
“FDA clearance of the self-collection kit marks a meaningful step toward removing today’s primary barriers to screening,” said Jianqing Bennett, senior vice president at Waters Advanced Diagnostics. “It supports more personalized care.”
Nationwide Rollout Planned Through Partnerships
Waters said it is building partnerships to expand nationwide access to the At-home HPV test screening kit, which will be available by prescription in the coming months. Patients will be able to receive the kit by mail, collect a sample at home, and send it to a laboratory for analysis.
Results will be shared with the patient’s health care provider, who can recommend follow-up care if needed. The company said the kit is expected to be covered by private insurance as well as government programs, including Medicaid and Medicare.
Health advocates say the initiative could help address disparities in cervical cancer outcomes, particularly among low-income and rural populations. By simplifying the screening process, officials hope more individuals will participate in routine testing.
The FDA’s decision marks a significant step in expanding access to preventive care and advancing efforts to reduce the burden of cervical cancer in the United States.